Abstract
Background
Most people with anorexia nervosa (pAN) have digestive symptoms, which affect tolerance to nutritional treatment. Oligomeric oral nutritional supplements may be better absorbed and tolerated than polymeric ones. Our aim was to compare an oligomeric versus a polymeric nutritional supplement in pAN in terms of worsening of digestive symptoms after ingestion (primary objective) and in terms of sensory perception of the supplement (secondary objective).
Methods
A randomised, crossover, double-blind, controlled clinical trial was conducted. Inclusion criteria were: male or female aged 12–50 years with anorexia nervosa treated at the Eating Disorders Unit of the Hospital Regional Universitario de Málaga, Spain. Participants were randomised to first receive one container of the oligomeric or polymeric supplement. A digestive symptom questionnaire was administered, which they completed again after taking the supplement, together with a questionnaire for sensory evaluation of the supplement. One week later, they received a container of the other type of supplement, and the process was repeated. Analysis was performed on a per protocol basis.
Results
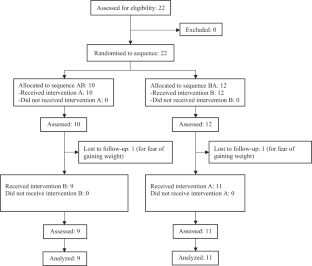
Ten people were randomised to the oligomeric-polymeric sequence and 12 to the polymeric-oligomeric sequence, of whom 9 and 11, respectively, finally received the intervention and were analysed. There were no statistically significant differences in digestive symptoms between the two supplements. The oligomeric one scored lower on taste and higher on acidity. The median (interquartile range) for oligomeric and polymeric were respectively: taste 6.5 (4), 8(3), p 0.03; acidity 0 (1), 0 (0), p 0.01.
Conclusions
In pAN, there is no difference in the worsening of digestive symptoms after ingestion of oligomeric versus polymeric supplements. Polymerics are perceived as having better taste and less acidity.
Trial registration
The trial is registered at ClinicalTrials.gov, number NCT05184738.
This is a preview of subscription content, access via your institution
Access options
/* style specs end */
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
/* style specs start */
style {
display: none !important;
}
.LiveAreaSection * {
align-content: stretch;
align-items: stretch;
align-self: auto;
animation-delay: 0s;
animation-direction: normal;
animation-duration: 0s;
animation-fill-mode: none;
animation-iteration-count: 1;
animation-name: none;
animation-play-state: running;
animation-timing-function: ease;
azimuth: center;
backface-visibility: visible;
background-attachment: scroll;
background-blend-mode: normal;
background-clip: borderBox;
background-color: transparent;
background-image: none;
background-origin: paddingBox;
background-position: 0 0;
background-repeat: repeat;
background-size: auto auto;
block-size: auto;
border-block-end-color: currentcolor;
border-block-end-style: none;
border-block-end-width: medium;
border-block-start-color: currentcolor;
border-block-start-style: none;
border-block-start-width: medium;
border-bottom-color: currentcolor;
border-bottom-left-radius: 0;
border-bottom-right-radius: 0;
border-bottom-style: none;
border-bottom-width: medium;
border-collapse: separate;
border-image-outset: 0s;
border-image-repeat: stretch;
border-image-slice: 100%;
border-image-source: none;
border-image-width: 1;
border-inline-end-color: currentcolor;
border-inline-end-style: none;
border-inline-end-width: medium;
border-inline-start-color: currentcolor;
border-inline-start-style: none;
border-inline-start-width: medium;
border-left-color: currentcolor;
border-left-style: none;
border-left-width: medium;
border-right-color: currentcolor;
border-right-style: none;
border-right-width: medium;
border-spacing: 0;
border-top-color: currentcolor;
border-top-left-radius: 0;
border-top-right-radius: 0;
border-top-style: none;
border-top-width: medium;
bottom: auto;
box-decoration-break: slice;
box-shadow: none;
box-sizing: border-box;
break-after: auto;
break-before: auto;
break-inside: auto;
caption-side: top;
caret-color: auto;
clear: none;
clip: auto;
clip-path: none;
color: initial;
column-count: auto;
column-fill: balance;
column-gap: normal;
column-rule-color: currentcolor;
column-rule-style: none;
column-rule-width: medium;
column-span: none;
column-width: auto;
content: normal;
counter-increment: none;
counter-reset: none;
cursor: auto;
display: inline;
empty-cells: show;
filter: none;
flex-basis: auto;
flex-direction: row;
flex-grow: 0;
flex-shrink: 1;
flex-wrap: nowrap;
float: none;
font-family: initial;
font-feature-settings: normal;
font-kerning: auto;
font-language-override: normal;
font-size: medium;
font-size-adjust: none;
font-stretch: normal;
font-style: normal;
font-synthesis: weight style;
font-variant: normal;
font-variant-alternates: normal;
font-variant-caps: normal;
font-variant-east-asian: normal;
font-variant-ligatures: normal;
font-variant-numeric: normal;
font-variant-position: normal;
font-weight: 400;
grid-auto-columns: auto;
grid-auto-flow: row;
grid-auto-rows: auto;
grid-column-end: auto;
grid-column-gap: 0;
grid-column-start: auto;
grid-row-end: auto;
grid-row-gap: 0;
grid-row-start: auto;
grid-template-areas: none;
grid-template-columns: none;
grid-template-rows: none;
height: auto;
hyphens: manual;
image-orientation: 0deg;
image-rendering: auto;
image-resolution: 1dppx;
ime-mode: auto;
inline-size: auto;
isolation: auto;
justify-content: flexStart;
left: auto;
letter-spacing: normal;
line-break: auto;
line-height: normal;
list-style-image: none;
list-style-position: outside;
list-style-type: disc;
margin-block-end: 0;
margin-block-start: 0;
margin-bottom: 0;
margin-inline-end: 0;
margin-inline-start: 0;
margin-left: 0;
margin-right: 0;
margin-top: 0;
mask-clip: borderBox;
mask-composite: add;
mask-image: none;
mask-mode: matchSource;
mask-origin: borderBox;
mask-position: 0 0;
mask-repeat: repeat;
mask-size: auto;
mask-type: luminance;
max-height: none;
max-width: none;
min-block-size: 0;
min-height: 0;
min-inline-size: 0;
min-width: 0;
mix-blend-mode: normal;
object-fit: fill;
object-position: 50% 50%;
offset-block-end: auto;
offset-block-start: auto;
offset-inline-end: auto;
offset-inline-start: auto;
opacity: 1;
order: 0;
orphans: 2;
outline-color: initial;
outline-offset: 0;
outline-style: none;
outline-width: medium;
overflow: visible;
overflow-wrap: normal;
overflow-x: visible;
overflow-y: visible;
padding-block-end: 0;
padding-block-start: 0;
padding-bottom: 0;
padding-inline-end: 0;
padding-inline-start: 0;
padding-left: 0;
padding-right: 0;
padding-top: 0;
page-break-after: auto;
page-break-before: auto;
page-break-inside: auto;
perspective: none;
perspective-origin: 50% 50%;
pointer-events: auto;
position: static;
quotes: initial;
resize: none;
right: auto;
ruby-align: spaceAround;
ruby-merge: separate;
ruby-position: over;
scroll-behavior: auto;
scroll-snap-coordinate: none;
scroll-snap-destination: 0 0;
scroll-snap-points-x: none;
scroll-snap-points-y: none;
scroll-snap-type: none;
shape-image-threshold: 0;
shape-margin: 0;
shape-outside: none;
tab-size: 8;
table-layout: auto;
text-align: initial;
text-align-last: auto;
text-combine-upright: none;
text-decoration-color: currentcolor;
text-decoration-line: none;
text-decoration-style: solid;
text-emphasis-color: currentcolor;
text-emphasis-position: over right;
text-emphasis-style: none;
text-indent: 0;
text-justify: auto;
text-orientation: mixed;
text-overflow: clip;
text-rendering: auto;
text-shadow: none;
text-transform: none;
text-underline-position: auto;
top: auto;
touch-action: auto;
transform: none;
transform-box: borderBox;
transform-origin: 50% 50%0;
transform-style: flat;
transition-delay: 0s;
transition-duration: 0s;
transition-property: all;
transition-timing-function: ease;
vertical-align: baseline;
visibility: visible;
white-space: normal;
widows: 2;
width: auto;
will-change: auto;
word-break: normal;
word-spacing: normal;
word-wrap: normal;
writing-mode: horizontalTb;
z-index: auto;
-webkit-appearance: none;
-moz-appearance: none;
-ms-appearance: none;
appearance: none;
margin: 0;
}
.LiveAreaSection {
width: 100%;
}
.LiveAreaSection .login-option-buybox {
display: block;
width: 100%;
font-size: 17px;
line-height: 30px;
color: #222;
padding-top: 30px;
font-family: Harding, Palatino, serif;
}
.LiveAreaSection .additional-access-options {
display: block;
font-weight: 700;
font-size: 17px;
line-height: 30px;
color: #222;
font-family: Harding, Palatino, serif;
}
.LiveAreaSection .additional-login > li:not(:first-child)::before {
transform: translateY(-50%);
content: “”;
height: 1rem;
position: absolute;
top: 50%;
left: 0;
border-left: 2px solid #999;
}
.LiveAreaSection .additional-login > li:not(:first-child) {
padding-left: 10px;
}
.LiveAreaSection .additional-login > li {
display: inline-block;
position: relative;
vertical-align: middle;
padding-right: 10px;
}
.BuyBoxSection {
display: flex;
flex-wrap: wrap;
flex: 1;
flex-direction: row-reverse;
margin: -30px -15px 0;
}
.BuyBoxSection .box-inner {
width: 100%;
height: 100%;
padding: 30px 5px;
display: flex;
flex-direction: column;
justify-content: space-between;
}
.BuyBoxSection p {
margin: 0;
}
.BuyBoxSection .readcube-buybox {
background-color: #f3f3f3;
flex-shrink: 1;
flex-grow: 1;
flex-basis: 255px;
background-clip: content-box;
padding: 0 15px;
margin-top: 30px;
}
.BuyBoxSection .subscribe-buybox {
background-color: #f3f3f3;
flex-shrink: 1;
flex-grow: 4;
flex-basis: 300px;
background-clip: content-box;
padding: 0 15px;
margin-top: 30px;
}
.BuyBoxSection .subscribe-buybox-nature-plus {
background-color: #f3f3f3;
flex-shrink: 1;
flex-grow: 4;
flex-basis: 100%;
background-clip: content-box;
padding: 0 15px;
margin-top: 30px;
}
.BuyBoxSection .title-readcube,
.BuyBoxSection .title-buybox {
display: block;
margin: 0;
margin-right: 10%;
margin-left: 10%;
font-size: 24px;
line-height: 32px;
color: #222;
text-align: center;
font-family: Harding, Palatino, serif;
}
.BuyBoxSection .title-asia-buybox {
display: block;
margin: 0;
margin-right: 5%;
margin-left: 5%;
font-size: 24px;
line-height: 32px;
color: #222;
text-align: center;
font-family: Harding, Palatino, serif;
}
.BuyBoxSection .asia-link,
.Link-328123652,
.Link-2926870917,
.Link-2291679238,
.Link-595459207 {
color: #069;
cursor: pointer;
text-decoration: none;
font-size: 1.05em;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
line-height: 1.05em6;
}
.BuyBoxSection .access-readcube {
display: block;
margin: 0;
margin-right: 10%;
margin-left: 10%;
font-size: 14px;
color: #222;
padding-top: 10px;
text-align: center;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
line-height: 20px;
}
.BuyBoxSection ul {
margin: 0;
}
.BuyBoxSection .link-usp {
display: list-item;
margin: 0;
margin-left: 20px;
padding-top: 6px;
list-style-position: inside;
}
.BuyBoxSection .link-usp span {
font-size: 14px;
color: #222;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
line-height: 20px;
}
.BuyBoxSection .access-asia-buybox {
display: block;
margin: 0;
margin-right: 5%;
margin-left: 5%;
font-size: 14px;
color: #222;
padding-top: 10px;
text-align: center;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
line-height: 20px;
}
.BuyBoxSection .access-buybox {
display: block;
margin: 0;
margin-right: 10%;
margin-left: 10%;
font-size: 14px;
color: #222;
opacity: 0.8px;
padding-top: 10px;
text-align: center;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
line-height: 20px;
}
.BuyBoxSection .price-buybox {
display: block;
font-size: 30px;
color: #222;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
padding-top: 30px;
text-align: center;
}
.BuyBoxSection .price-buybox-to {
display: block;
font-size: 30px;
color: #222;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
text-align: center;
}
.BuyBoxSection .price-info-text {
font-size: 16px;
padding-right: 10px;
color: #222;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
}
.BuyBoxSection .price-value {
font-size: 30px;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
}
.BuyBoxSection .price-per-period {
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
}
.BuyBoxSection .price-from {
font-size: 14px;
padding-right: 10px;
color: #222;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
line-height: 20px;
}
.BuyBoxSection .issue-buybox {
display: block;
font-size: 13px;
text-align: center;
color: #222;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
line-height: 19px;
}
.BuyBoxSection .no-price-buybox {
display: block;
font-size: 13px;
line-height: 18px;
text-align: center;
padding-right: 10%;
padding-left: 10%;
padding-bottom: 20px;
padding-top: 30px;
color: #222;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
}
.BuyBoxSection .vat-buybox {
display: block;
margin-top: 5px;
margin-right: 20%;
margin-left: 20%;
font-size: 11px;
color: #222;
padding-top: 10px;
padding-bottom: 15px;
text-align: center;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
line-height: 17px;
}
.BuyBoxSection .tax-buybox {
display: block;
width: 100%;
color: #222;
padding: 20px 16px;
text-align: center;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
line-height: NaNpx;
}
.BuyBoxSection .button-container {
display: flex;
padding-right: 20px;
padding-left: 20px;
justify-content: center;
}
.BuyBoxSection .button-container > * {
flex: 1px;
}
.BuyBoxSection .button-container > a:hover,
.Button-505204839:hover,
.Button-1078489254:hover,
.Button-2737859108:hover {
text-decoration: none;
}
.BuyBoxSection .btn-secondary {
background: #fff;
}
.BuyBoxSection .button-asia {
background: #069;
border: 1px solid #069;
border-radius: 0;
cursor: pointer;
display: block;
padding: 9px;
outline: 0;
text-align: center;
text-decoration: none;
min-width: 80px;
margin-top: 75px;
}
.BuyBoxSection .button-label-asia,
.ButtonLabel-3869432492,
.ButtonLabel-3296148077,
.ButtonLabel-1636778223 {
display: block;
color: #fff;
font-size: 17px;
line-height: 20px;
font-family: -apple-system, BlinkMacSystemFont, “Segoe UI”, Roboto,
Oxygen-Sans, Ubuntu, Cantarell, “Helvetica Neue”, sans-serif;
text-align: center;
text-decoration: none;
cursor: pointer;
}
.Button-505204839,
.Button-1078489254,
.Button-2737859108 {
background: #069;
border: 1px solid #069;
border-radius: 0;
cursor: pointer;
display: block;
padding: 9px;
outline: 0;
text-align: center;
text-decoration: none;
min-width: 80px;
max-width: 320px;
margin-top: 20px;
}
.Button-505204839 .btn-secondary-label,
.Button-1078489254 .btn-secondary-label,
.Button-2737859108 .btn-secondary-label {
color: #069;
}
.uList-2102244549 {
list-style: none;
padding: 0;
margin: 0;
}
/* style specs end */

Data availability
Data supporting the results of this trial are available from the corresponding author upon reasonable request.
References
-
Jafar W, Morgan J. Anorexia nervosa and the gastrointestinal tract. Frontline Gastroenterol. 2022;13:316–24.
Google Scholar
-
Schalla MA, Stengel A. Gastrointestinal alterations in anorexia nervosa—a systematic review. Eur Eat Disord Rev J Eat Disord Assoc. 2019;27:447–61.
-
Kamal N, Chami T, Andersen A, Rosell FA, Schuster MM, Whitehead WE. Delayed gastrointestinal transit times in anorexia nervosa and bulimia nervosa. Gastroenterology. 1991;101:1320–4.
Google Scholar
-
Hirakawa M, Okada T, Iida M, Tamai H, Kobayashi N, Nakagawa T, et al. Small bowel transit time measured by hydrogen breath test in patients with anorexia nervosa. Dig Dis Sci. 1990;35:733–6.
Google Scholar
-
Mehler PS, Brown C. Anorexia nervosa—medical complications. J Eat Disord. 2015;3:11.
Google Scholar
-
Rigaud D, Bedig G, Merrouche M, Vulpillat M, Bonfils S, Apfelbaum M. Delayed gastric emptying in anorexia nervosa is improved by completion of a renutrition program. Dig Dis Sci. 1988;33:919–25.
Google Scholar
-
Waldholtz BD, Andersen AE. Gastrointestinal symptoms in anorexia nervosa. A prospective study. Gastroenterology. 1990;98:1415–9.
Google Scholar
-
Chiarioni G, Bassotti G, Monsignori A, Menegotti M, Salandini L, Di Matteo G, et al. Anorectal dysfunction in constipated women with anorexia nervosa. Mayo Clin Proc. 2000;75:1015–9.
Google Scholar
-
Szmukler GI, Young GP, Lichtenstein M, Andrews JT. A serial study of gastric emptying in anorexia nervosa and bulimia. Aust N Z J Med. 1990;20:220–5.
Google Scholar
-
West M, McMaster CM, Staudacher HM, Hart S, Jacka FN, Stewart T, et al. Gastrointestinal symptoms following treatment for anorexia nervosa: a systematic literature review. Int J Eat Disord. 2021;54:936–51.
Google Scholar
-
Golden NH, Meyer W. Nutritional rehabilitation of anorexia nervosa. Goals and dangers. Int J Adolesc Med Health. 2004;16:131–44.
Google Scholar
-
Gómez-Candela C, Palma Milla S, Miján-de-la-Torre A, Rodríguez Ortega P, Matía Martín P, Loria Kohen V, et al. [Consensus document about the nutritional evaluation and management of eating disorders: anorexia nervosa]. Nutr Hosp. 2018;35:11–48.
Google Scholar
-
Marzola E, Nasser JA, Hashim SA, Shih PAB, Kaye WH. Nutritional rehabilitation in anorexia nervosa: review of the literature and implications for treatment. BMC Psychiatry. 2013;13:1–13.
-
Gómez Candela C, Palma Milla S, Miján-de-la-Torre A, Rodríguez Ortega P, Matía Martín P, Loria Cohen V, et al. [Consensus document about the nutritional evaluation and management of eating disorders: bulimia nervosa, binge eating disorder, and others]. Nutr Hosp. 2018;35:49–97.
Google Scholar
-
Imbierowicz K, Braks K, Jacoby GE, Geiser F, Conrad R, Schilling G, et al. High-caloric supplements in anorexia treatment. Int J Eat Disord. 2002;32:135–45.
Google Scholar
-
Olveira Fuster G, editor. Manual de nutrición clínica y dietética. 4th ed. Málaga: Díaz de Santos; 2023.
-
Sanz-Paris A, Martinez-García M, Martinez-Trufero J, Lambea-Sorrosal J, Calvo-Gracia F, López-Alaminos ME. Oligomeric enteral nutrition in undernutrition, due to oncology treatment-related diarrhea. Systematic review and proposal of an algorithm of action. Nutrients. 2019;11:1888.
Google Scholar
-
American Psychiatric Association. Diagnostic and statistical manual of mental disorders, Fifth Edition (DSM-5). Arlington: American Psychiatric Association; 2013.
-
Cuerda C, Pironi L, Arends J, Bozzetti F, Gillanders L, Jeppesen PB, et al. ESPEN practical guideline: clinical nutrition in chronic intestinal failure. Clin Nutr. 2021;40:5196–220.
Google Scholar
-
Arvanitakis M, Ockenga J, Bezmarevic M, Gianotti L, Krznarić Ž, Lobo DN, et al. ESPEN guideline on clinical nutrition in acute and chronic pancreatitis. Clin Nutr. 2020;39:612–31.
Google Scholar
-
Nakahigashi M, Yamamoto T, Sacco R, Hanai H, Kobayashi F. Enteral nutrition for maintaining remission in patients with quiescent Crohn’s disease: current status and future perspectives. Int J Colorectal Dis. 2016;31:1–7.
Google Scholar
-
Akobeng AK, Thomas AG. Enteral nutrition for maintenance of remission in Crohn’s disease. Cochrane Database Syst Rev. 2007;18:CD005984.
-
Petrov MS, Loveday BPT, Pylypchuk RD, McIlroy K, Phillips ARJ, Windsor JA. Systematic review and meta-analysis of enteral nutrition formulations in acute pancreatitis. Br J Surg. 2009;96:1243–52.
Google Scholar
-
Mukai J, Miyanaga Y, Ishizaka T, Asaka K, Nakai Y, Tsuji E, et al. Quantitative taste evaluation of total enteral nutrients. Chem Pharm Bull. 2004;52:1416–21.
Google Scholar
-
McGough C, Peacock N, Hackett C, Baldwin C, Norman A, Frost G, et al. Taste preferences for oral nutrition supplements in patients before and after pelvic radiotherapy: a double-blind controlled study. Clin Nutr. 2006;25:906–12.
Google Scholar
-
Marzola E, Cavallo F, Pradella P, Brustolin A, Abbate-Daga G. A tasting experiment comparing food and nutritional supplement in anorexia nervosa. Appetite. 2020;155:104789.
Google Scholar
-
Trovato CM, Capriati T, Bolasco G, Campana C, Papa V, Mazzoli B, et al. Five-year inpatient management of teenagers with anorexia nervosa: focus on nutritional issues. J Pediatr Gastroenterol Nutr. 2022;74:674–80.
Google Scholar
Acknowledgements
The study was monitored by the METAnetwork. The IBIMA Department of Biostatistics collaborated in the statistical analysis of the study.
Funding
Adventia Pharma partially collaborated with an unconditional grant and the free provision of nutritional supplements. IRG holds a Río Hortega contract from the Carlos III National Health Institute, co-funded by European Social Fund 2014-2020 “The FSE invests in your future”; number: CM20/00225. The researchers declare their independence in the conduct of the study. The funder had no role in the study design, data collection, analysis, interpretation of data, the writing of this article, or the decision to submit it for publication.
Author information
Authors and Affiliations
Contributions
Conceptualization: GO; methodology: IRG, FGT, NPP, GO; software: IRG, NPP, GO; validation: IRG, GO; formal analysis: IRG, NPP, FJST, GO; investigation: IRG, FGT, NPP, CFM, LOD, GO; resources: IRG, FGT, NPP, CFM, LOD, FJST, MPL, GO; data curation: IRG, NPP; writing—original draft preparation: IRG; writing—review and editing: IRG, GO; visualization: IRG, GO; supervision: IRG, GO; project administration: GO; funding acquisition: IRG, GO. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
IRG has received honorarium support for a presentation from Adventia Pharma. GO has received occasional honorarium support for presentations, meeting attendance, and travel from Adventia Pharma. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationship that could be construed as a potential conflict of interest.
Ethics approval and consent to participate
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Malaga Provincial Research Ethics Committee on 30 September 2021, code 1735-N-21. Informed consent was obtained from all subjects included in the study. The material used in this article is in the public domain or is under an open licence that allows its reproduction. Every effort has been made to properly attribute the original source of the material used.
Consent for publication
Informed consent was obtained from all subjects involved in the study. In the case of minors, the informed consent was signed by their legal representative.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Reprints and permissions
About this article
Cite this article
Ruiz García, I., Porras Pérez, N., García Torres, F. et al. Comparison of the digestive tolerance of an oligomeric versus a polymeric oral nutritional supplement in people with anorexia nervosa. A randomised crossover clinical trial.
Eur J Clin Nutr (2025). https://doi.org/10.1038/s41430-025-01608-y
-
Received: 19 December 2023
-
Revised: 13 February 2025
-
Accepted: 18 March 2025
-
Published: 31 March 2025
-
DOI: https://doi.org/10.1038/s41430-025-01608-y