Abstract
The causal roles of muscle weakness in cardiometabolic diseases and osteoporosis remain elusive. This two-sample Mendelian randomization (MR) study aims to explore the causal roles of muscle weakness in the risk of cardiometabolic diseases and osteoporosis. 15 single nucleotide polymorphisms (SNPs, P < 5 × 10−8) associated with muscle weakness were used as instrumental variables. Genetic predisposition to muscle weakness led to increased risk of coronary artery disease (inverse variance weighted [IVW] analysis, beta-estimate: 0.095, 95% confidence interval [CI]: 0.023 to 0.166, standard error [SE]:0.036, P-value = 0.009) and reduced risk of heart failure (weight median analysis, beta-estimate: − 0.137, 95% CI − 0.264 to − 0.009, SE:0.065, P-value = 0.036). In addition, muscle weakness may reduce the estimated bone mineral density (eBMD, weight median analysis, beta-estimate: − 0.059, 95% CI − 0.110 to − 0.008, SE:0.026, P-value = 0.023). We found no MR associations between muscle weakness and atrial fibrillation, type 2 diabetes or fracture. This study provides robust evidence that muscle weakness is causally associated with the incidence of coronary artery disease and heart failure, which may provide new insight to prevent and treat these two cardiometabolic diseases.
Introduction
Muscle weakness commonly occurs as the advancing age and it is a fundamental component of frailty and sarcopenia1,2,3. Compared to individuals in twenties, population with over 70 years are estimated to suffer from up to 20% lost muscle mass4. Loss of muscle mass (sarcopenia) is closely associated with muscle weakness which may affect health outcomes5,6. Patients with muscle weakness commonly have some difficulties in daily activity and low muscle strength as measured by hand grip strength, which has become a predictive factor of morbidity and mortality4,7. Muscle weakness is heritable and can be used for genetic studies8.
Several observational studies reported that muscle weakness had some association with the incidence of cardiometabolic diseases and osteoporosis, but these results are conflicting9,10,11,12,13,14,15. Potential confounding factors and reverse causality in these studies may affect the association between muscle weakness and cardiometabolic diseases/osteoporosis. Cardiometabolic diseases and osteoporosis are also highly polygenic traits based on the results of genome-wide association studies (GWASs)16,17,18,19,20,21,22.
Mendelian randomization (MR) study is widely used to establish the causal relationship between exposure phenotype and outcome phenotype, with the advantages of preventing reverse causation and potential confounding factors23,24,25,26,27. Furthermore, the two-sample MR study is able to increase the scope and statistical power of MR25,28,29,30,31. Due to the high heritability of muscle weakness, cardiometabolic diseases and osteoporosis, this two-sample MR study aims to explore the causal influence of muscle weakness on the incidence of cardiometabolic diseases and osteoporosis.
Methods
Genetic instrument for muscle weakness
The largest available GWAS meta-analysis included 22 independent cohorts with maximum hand grip strength recorded (i.e. the UK Biobank, the US Health and Retirement Study, the Framingham Heart Study, and others) and total 256,523 individuals of European descent aged 60 years or older. Among them, 46,596 participants was diagnosed with muscle weakness based on hand grip strength and EWGSOP definition: grip strength < 30 kg for male individuals and < 20 kg for female individuals32.
Initially, 15 single nucleotide polymorphisms (SNPs) showed robust association with muscle weakness (P < 5 × 10−8). Linkage disequilibrium (LD) between selected SNPs was calculated using European samples from the 1000 Genomes project. No SNPs were excluded due to high LD (r2 ≥ 0.001). Finally, 15 SNPs were used as instrumental variables (Supplementary Table 1). The proxy SNPs in linkage disequilibrium (LD, r2 > 0.9) were used if original SNPs were unavailable in the outcome database. Thus, rs6488725 was used as the proxy for rs34464763 among all outcomes (Supplementary Table 2).
Outcome data sources
The genetic associations of each outcome from GWASs were presented in Table 1. Briefly, we included the GWAS summary data of cardiometabolic diseases including coronary artery disease (547,261 individuals) from UK Biobank and CARDIoGRAMplusC4D33, heart failure (977,323 individuals) from UK Biobank34, atrial fibrillation (587,446 individuals) from one large meta-analysis35 and type 2 diabetes (898,130 individuals) from DIAGRAM36. In terms of osteoporosis and fracture, the outcome measures included bone mineral density (BMD) as estimated by heel quantitative ultrasound (eBMD) and fracture among 426,824 people. Fracture cases were defined as any fracture apart from the fracture of skull, face, hands, feet, pathological fractures due to malignancy, atypical femoral fractures, periprosthetic and healed fracture37. Most GWASs were adjusted for sex, body mass index (BMI) and genetic principal components. All participants were all from European descent except for those with atrial fibrillation from predominant European descent (mixed descents). Supplementary Table 2 showed the summary statistics for the SNPs related to muscle weakness and corresponding statistics of outcomes.
Statistical analyses
To determine causal influence of muscle weakness on each outcome, we conducted the inverse variance weighted (IVW) analysis because more than 2 SNPs were available. IVW method used a meta-analysis approach to combine Wald estimates for each SNP in order to get the overall estimates of the effect of muscle weakness on each outcome23. The weighted median and MR-Egger regression methods were also applied to estimate the effects. Cochrane’s Q-statistic was used to assess the heterogeneity of SNP effects and P < 0.05 indicated significant heterogeneity38. MR pleiotropy residual sum and outlier test (MR-PRESSO) aimed to assess the presence of pleiotropy and the effect estimates were recalculated after outlying SNPs were excluded39.
All methods were carried out in accordance with relevant guidelines and regulations. All experimental protocols were approved and the ethical approval for each study can be found in the original publications (including informed consent from each participant). P < 0.05 indicated statistical difference. All of these analyses were conducted in R V.4.0.4 by using the R packages of ‘MendelianRandomization’40, ‘TwoSampleMR’41 and ‘MR-PRESSO’42.
Ethical approval
The ethical approval for each study included in this investigation can be found in the original publications.
Results
Cardiometabolic diseases
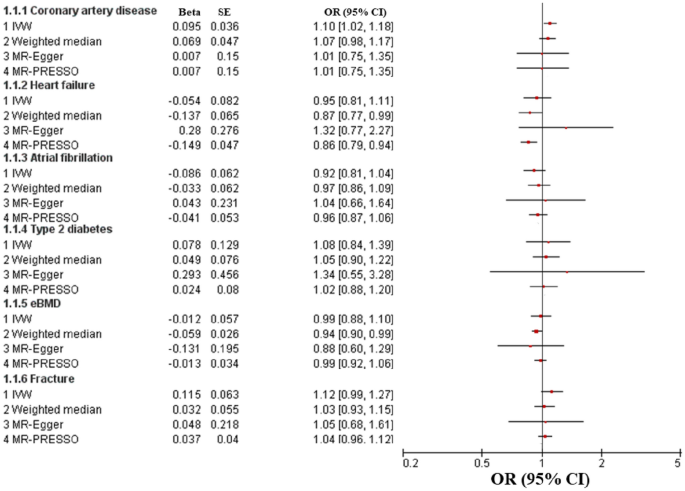
We evaluated the causal effect of muscle weakness on coronary artery disease, heart failure, atrial fibrillation and type 2 diabetes in this MR analysis (Table 2). IVW analysis demonstrated that genetically muscle weakness played a significant causal role in the increased risk of coronary artery disease (beta-estimate: 0.095, 95% CI 0.023 to 0.166, SE:0.036, P-value = 0.009), but it was not supported by the weighted-median analysis (beta-estimate: 0.069, 95% CI − 0.023 to 0.161, SE:0.047, P-value = 0.141, Fig. 1).

OR (95% CI) for causal association between muscle weakness and each outcome through multiple analyses.
According to weighted-median analysis, muscle weakness showed substantially causal effect on the reduced incidence of heart failure (beta-estimate: − 0.137, 95% CI − 0.264 to − 0.009, SE:0.065, P-value = 0.036), but it was not confirmed in the IVW analysis (beta-estimate: − 0.054, 95% CI − 0.214 to 0.106, SE:0.082, P-value = 0.506, Fig. 1). In addition, IVW analyses found that muscle weakness demonstrated no remarkable MR association with atrial fibrillation (beta-estimate: − 0.086, 95% CI − 0.207 to 0.035, SE:0.062, P-value = 0.162) or type 2 diabetes (beta-estimate: 0.078, 95% CI − 0.175 to 0.331, SE:0.129, P-value = 0.547), which were also confirmed by weighted-median analyses (Fig. 1).
Osteoporosis
This MR analysis also included outcome measures of eBMD and fracture (Table 2). According to weighted-median analysis, muscle weakness was casually associated with decreased eBMD (beta-estimate: − 0.059, 95% CI − 0.110 to − 0.008, SE:0.026, P-value = 0.023), but it was not supported by the IVW analysis (beta-estimate: − 0.012, 95% CI − 0.123 to 0.099, SE:0.057, P-value = 0.827, Fig. 1). Muscle weakness revealed no causal influence on fracture by IVW analysis (beta-estimate: 0.115, 95% CI − 0.008 to 0.238, SE:0.063, P-value = 0.066) or weighted-median analysis (beta-estimate: 0.032, 95% CI − 0.075 to 0.139, SE:0.055, P-value = 0.561, Fig. 1).
Evaluation of assumptions and sensitivity analyses
Little evidence of directional pleiotropy was found for all models (MR-Egger intercept P-value > 0.05, Table 2). There was significant heterogeneity for heart failure, atrial fibrillation, type 2 diabetes, eBMD and fracture. Thus, among the 15 SNP instrumental variables associated with muscle weakness, MR-PRESSO method identified 2 outliers (rs13107325, rs10952289) for heart failure, one outlier (rs143384) for atrial fibrillation, four outliers (rs7624084, rs34415150, rs10952289, rs62102286) for type 2 diabetes, 11 outliers (rs12140813, rs958685, rs7624084, rs13107325, rs34415150, rs10952289, rs11236213, rs34464763, rs3118903, rs8061064, rs62102286) for eBMD and 2 outliers (rs10952289, rs34464763) fracture (Table 3).
After excluding these outlying SNP variants, these remarkable MR associations were confirmed between muscle weakness and increased risk of coronary artery disease (Fig. 1 and Table 3). In addition, muscle weakness was confirmed to have a causal effect on low risk of heart failure (beta-estimate: − 0.149, 95% CI − 0.241 to − 0.056, SE:0.047, P-value = 0.002, Fig. 1 and Table 3). The MR association between muscle weakness with other outcomes were not changed after excluding the outlying SNP variants (Table 3).
Discussion
Our two-sample MR study found the robustly causal effect of muscle weakness on increased risk of coronary artery disease and decreased risk of heart failure, and these strong MR associations were confirmed by the sensitivity analyses. These positive findings indicated that the regulatory mechanisms of muscle weakness may provide new insight to prevent and treat coronary artery disease and heart failure. In addition, muscle weakness may have a causal role in reduced eBMD. We found no causal effect of muscle weakness on atrial fibrillation, type 2 diabetes or fracture.
Several observational studies and meta-analysis explored the association between muscle results and cardiometabolic diseases, but no conclusive results were found9,13,43. One meta-analysis revealed that handgrip strength was an independent predictor of cardiometabolic diseases in community-dwelling populations, but this this association was not significant after adjusting for baseline risk factors9,43. One recent MR analysis found no causality in the association between handgrip strength (European population) and coronary artery disease (mixed population). The large-scale genetic discovery analysis identified 16 loci associated with grip strength (P < 5 × 10−8) among 195,180 individuals as instrumental variables10, and that MR study included GWAS summary data related to coronary heart disease among 184,305 individuals44.
Our large-scale MR study was performed in larger populations including 256,523 individuals of European descent for muscle weakness and 547,261 individuals of European descent for coronary artery disease. Totally, 15 loci associated with grip strength (P < 5 × 10−8) were used as instrumental variables. The results provided the robust evidence for the causal association between muscle weakness and increased risk of coronary artery disease, which was confirmed by multiple sensitivity analyses. Muscle weakness and low muscle mass reduces total energy expenditure, which may result in high fat mass. Accumulated body fat mass triggers chronic inflammation, and is thought to be a risk factor for the development and progression of coronary artery disease45,46,47.
One leading cause of heart failure is coronary artery disease, but heart failure can be also caused by arrhythmias, hypertension, type 2 diabetes mellitus, obesity, and lifestyle factors (such as smoking). A large-scale observational study found that higher hand grip strength was independently associated with lower incidence of heart failure48. On the contrary, our MR study revealed that muscle weakness was causally associated with lower incidence of heart failure, which was confirm by the IVW analysis after excluding the outlying SNPs (beta-estimate: − 0.149, 95% CI − 0.241 to − 0.056, SE:0.047, P-value = 0.002, Fig. 1 and Table 3). This positive finding was very interesting, and may be attributed by the atrophy of the muscle fibers and reduced requirement of cardiac output due to low muscle mass49.
Patients with osteoporosis typically have the features of low bone mass, BMD and bone strength, which can increase the risk of fracture50,51,52,53,54. Several observational studies revealed the significant correlation between low grip strength and low BMD of the bones adjacent to the muscles related to grip55,56,57. In 1,168 menopausal women, Osei-Hyiaman et al. found the significant relationship between grip strength and BMD of metacarpal index55. Hasegawa et al. revealed that BMD of the distal radius was more associated with hand grip strength than with cross-sectional muscle area57. In contrast, Zimmermann et al. documented that hand grip strength in postmenopausal women showed no impact on vertebral BMD, but only affected femur BMD58, while Foley et al. documented no correlation between hand grip strength and femoral BMD59.
Considering these insistent results, our MR analyses revealed that muscle weakness may have a causal role in reduced eBMD. It is postulated that muscle contraction force provides a mechanical stress on the bones, which is accepted as an important osteogenic stimulus. There is bi-directional bone-muscle crosstalk, which is probably mediated by cytokines, osteokines, myokines, and other growth factors60. In addition, low BMD associated with muscle weakness may be associated with systemic inflammation and oxidative stress61,62.
Our results demonstrated that genetically muscle weakness was unlikely to be causally associated with atrial fibrillation, type 2 diabetes or fracture. The potential causal effect of muscle weakness to reduce eBMD was not translated to affect the risk of fracture. This two-sample MR study aims to investigate the causal effect of muscle weakness on the risk of cardiometabolic diseases and osteoporosis, and has the advantage of preventing reverse causation and confounding factors. The intercepts for the MR-Egger analysis suggest no directional pleiotropy for all outcomes. However, several limitations should be taken into consideration. Firstly, all the included participants are of predominantly European, and we can not directly apply our findings for other populations. Secondly, GWAS summary statistics can not be used to conduct MR analysis based on different age stratums. Thirdly, the contribution of muscle weakness to low eBMD is not translated to increased incidence of fracture, but the detail mechanisms are unclear.
Conclusion
This two-sample MR study provides strong evidence to confirm that muscle weakness is a significantly causal factor for increased risk of coronary artery disease and reduced risk of heart failure, and the related mechanisms may help prevent and treat these two diseases.
Data availability
Data supporting the findings of this study were available within the paper.
References
-
Owen, A. M. et al. Chronic muscle weakness and mitochondrial dysfunction in the absence of sustained atrophy in a preclinical sepsis model. Elife https://doi.org/10.7554/eLife.49920 (2019).
Google Scholar
-
Clark, B. C. & Manini, T. M. What is dynapenia?. Nutrition 28(5), 495–503 (2012).
Google Scholar
-
Manini, T. M. & Clark, B. C. Dynapenia and aging: An update. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 67(1), 28–40 (2012).
Google Scholar
-
Mitchell, W. K. et al. Sarcopenia, dynapenia, and the impact of advancing age on human skeletal muscle size and strength; a quantitative review. Front. Physiol. 3, 260 (2012).
Google Scholar
-
Cruz-Jentoft, A. J. & Sayer, A. A. Sarcopenia. Lancet 393(10191), 2636–2646 (2019).
Google Scholar
-
Cawthon, P. M. et al. Establishing the link between lean mass and grip strength cut points with mobility disability and other health outcomes: Proceedings of the sarcopenia definition and outcomes consortium conference. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 75(7), 1317–1323 (2020).
Google Scholar
-
Beaudart, C. et al. Sarcopenia in daily practice: Assessment and management. BMC Geriatr. 16(1), 170 (2016).
Google Scholar
-
Frederiksen, H. et al. Hand grip strength: A phenotype suitable for identifying genetic variants affecting mid- and late-life physical functioning. Genet. Epidemiol. 23(2), 110–122 (2002).
Google Scholar
-
Wu, Y., Wang, W., Liu, T. & Zhang, D. Association of grip strength with risk of all-cause mortality, cardiovascular diseases, and cancer in community-dwelling populations: A meta-analysis of prospective cohort studies. J. Am. Med. Dir. Assoc. 18(6), 551.e17-551.e35 (2017).
Google Scholar
-
Willems, S. M. et al. Large-scale GWAS identifies multiple loci for hand grip strength providing biological insights into muscular fitness. Nat. Commun. 8, 16015 (2017).
Google Scholar
-
Curcio, F. et al. Sarcopenia and heart failure. Nutrients 12(1), 211 (2020).
Google Scholar
-
Canteri, A. L. et al. Sarcopenia in heart failure with reduced ejection fraction. Am. J. Cardiovasc. Dis. 9(6), 116–126 (2019).
Google Scholar
-
Ko, B. J. et al. Relationship between low relative muscle mass and coronary artery calcification in healthy adults. Arterioscler. Thromb. Vasc. Biol. 36(5), 1016–1021 (2016).
Google Scholar
-
Grill, B., Levangie, P. K., Cole, M., Rosenberg, D. & Jensen, L. Bone mineral density among individuals with residual lower limb weakness after polio. PM & R 11(5), 470–475 (2019).
Google Scholar
-
Eguchi, Y. et al. Reduced leg muscle mass and lower grip strength in women are associated with osteoporotic vertebral compression fractures. Arch. Osteoporos. 14(1), 112 (2019).
Google Scholar
-
Warrington, N. M. et al. Maternal and fetal genetic effects on birth weight and their relevance to cardio-metabolic risk factors. Nat. Genet. 51(5), 804–814 (2019).
Google Scholar
-
Nelson, C. P. et al. Association analyses based on false discovery rate implicate new loci for coronary artery disease. Nat. Genet. 49(9), 1385–1391 (2017).
Google Scholar
-
Iotchkova, V. et al. Discovery and refinement of genetic loci associated with cardiometabolic risk using dense imputation maps. Nat. Genet. 48(11), 1303–1312 (2016).
Google Scholar
-
Zhou, W. et al. Efficiently controlling for case-control imbalance and sample relatedness in large-scale genetic association studies. Nat. Genet. 50(9), 1335–1341 (2018).
Google Scholar
-
Richards, J. B., Zheng, H. F. & Spector, T. D. Genetics of osteoporosis from genome-wide association studies: Advances and challenges. Nat. Rev. Genet. 13(8), 576–588 (2012).
Google Scholar
-
Trajanoska, K. & Rivadeneira, F. The genetic architecture of osteoporosis and fracture risk. Bone 126, 2–10 (2019).
Google Scholar
-
Yang, T. L. et al. A road map for understanding molecular and genetic determinants of osteoporosis. Nat. Rev. Endocrinol. 16(2), 91–103 (2020).
Google Scholar
-
Burgess, S., Dudbridge, F. & Thompson, S. G. Combining information on multiple instrumental variables in Mendelian randomization: Comparison of allele score and summarized data methods. Stat. Med. 35(11), 1880–1906 (2016).
Google Scholar
-
Dalbeth, N. et al. Mendelian randomization analysis to examine for a causal effect of urate on bone mineral density. J. Bone Miner. Res. 30(6), 985–991 (2015).
Google Scholar
-
He, B. et al. Depression and osteoporosis: A Mendelian randomization study. Calcif. Tissue Int. 109(6), 675–684 (2021).
Google Scholar
-
Larsson, S. C., Traylor, M. & Markus, H. S. Circulating vitamin K1 levels in relation to ischemic stroke and its subtypes: A Mendelian randomization study. Nutrients 10(11), 1575 (2018).
Google Scholar
-
Zhao, J. et al. Systematic influence of circulating bilirubin levels on osteoporosis. Front. Endocrinol. https://doi.org/10.3389/fendo.2021.719920 (2021).
Google Scholar
-
Pierce, B. L. & Burgess, S. Efficient design for Mendelian randomization studies: Subsample and 2-sample instrumental variable estimators. Am. J. Epidemiol. 178(7), 1177–1184 (2013).
Google Scholar
-
Burgess, S., Scott, R. A., Timpson, N. J., Davey Smith, G. & Thompson, S. G. Using published data in Mendelian randomization: A blueprint for efficient identification of causal risk factors. Eur. J. Epidemiol. 30(7), 543–552 (2015).
Google Scholar
-
He, B. et al. Causal roles of circulating adiponectin in osteoporosis and cancers. Bone 155, 116266 (2022).
Google Scholar
-
He, B. et al. Causal roles of sleep duration in osteoporosis and cardiometabolic diseases: A Mendelian randomization study. BioMed Res. Int. 2022, 6819644 (2022).
Google Scholar
-
Jones, G. et al. Genome-wide meta-analysis of muscle weakness identifies 15 susceptibility loci in older men and women. Nat. Commun. 12(1), 654 (2021).
Google Scholar
-
van der Harst, P. & Verweij, N. Identification of 64 novel genetic loci provides an expanded view on the genetic architecture of coronary artery disease. Circ. Res. 122(3), 433–443 (2018).
Google Scholar
-
Shah, S. et al. Genome-wide association and Mendelian randomisation analysis provide insights into the pathogenesis of heart failure. Nat. Commun. 11(1), 163 (2020).
Google Scholar
-
Roselli, C. et al. Multi-ethnic genome-wide association study for atrial fibrillation. Nat. Genet. 50(9), 1225–1233 (2018).
Google Scholar
-
Mahajan, A. et al. Fine-mapping type 2 diabetes loci to single-variant resolution using high-density imputation and islet-specific epigenome maps. Nat. Genet. 50(11), 1505–1513 (2018).
Google Scholar
-
Morris, J. A. et al. An atlas of genetic influences on osteoporosis in humans and mice. Nat. Genet. 51(2), 258–266 (2019).
Google Scholar
-
Bowden, J., Hemani, G. & Davey Smith, G. Invited commentary: Detecting individual and global horizontal pleiotropy in mendelian randomization—A job for the humble heterogeneity statistic?. Am. J. Epidemiol. 187(12), 2681–2685 (2018).
Google Scholar
-
Burgess, S. & Thompson, S. G. Multivariable Mendelian randomization: The use of pleiotropic genetic variants to estimate causal effects. Am. J. Epidemiol. 181(4), 251–260 (2015).
Google Scholar
-
Yavorska, O. O. & Burgess, S. MendelianRandomization: An R package for performing Mendelian randomization analyses using summarized data. Int. J. Epidemiol. 46(6), 1734–1739 (2017).
Google Scholar
-
Hemani, G. et al. The MR-Base platform supports systematic causal inference across the human phenome. eLife 7, e34408 (2018).
Google Scholar
-
Verbanck, M., Chen, C. Y., Neale, B. & Do, R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat. Genet. 50(5), 693–698 (2018).
Google Scholar
-
Gubelmann, C., Vollenweider, P. & Marques-Vidal, P. No association between grip strength and cardiovascular risk: The CoLaus population-based study. Int. J. Cardiol. 236, 478–482 (2017).
Google Scholar
-
Nikpay, M. et al. A comprehensive 1,000 Genomes-based genome-wide association meta-analysis of coronary artery disease. Nat. Genet. 47(10), 1121–1130 (2015).
Google Scholar
-
Lee, K. Muscle mass and body fat in relation to cardiovascular risk estimation and lipid-lowering eligibility. J. Clin. Densitom. 20(2), 247–255 (2017).
Google Scholar
-
Kim, T. N. & Choi, K. M. The implications of sarcopenia and sarcopenic obesity on cardiometabolic disease. J. Cell. Biochem. 116(7), 1171–1178 (2015).
Google Scholar
-
Pearson, T. A. et al. Markers of inflammation and cardiovascular disease: Application to clinical and public health practice: A statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Circulation 107(3), 499–511 (2003).
Google Scholar
-
Sillars, A. et al. Association of fitness and grip strength with heart failure: Findings from the UK Biobank population-based study. Mayo Clin. Proc. 94(11), 2230–2240 (2019).
Google Scholar
-
Beyer, S. E. et al. Prospective association between handgrip strength and cardiac structure and function in UK adults. PLoS ONE 13(3), e0193124 (2018).
Google Scholar
-
Ensrud, K. E. & Crandall, C. J. Osteoporosis. Ann. Intern. Med. 167(3), ITC17–ITC32 (2017).
Google Scholar
-
Kanis, J. A., Cooper, C., Rizzoli, R. & Reginster, J. Y. European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos. Int. 30(1), 3–44 (2019).
Google Scholar
-
Zhang, M. et al. Causal associations of circulating adiponectin with cardiometabolic diseases and osteoporotic fracture. Sci. Rep. 12(1), 6689 (2022).
Google Scholar
-
He, B. et al. Comparison of necroptosis with apoptosis for OVX-induced osteoporosis. Front. Mol. Biosci. 8(1262), 790613 (2021).
Google Scholar
-
He, B. et al. Causal effect of blood pressure on bone mineral density and fracture: A Mendelian randomization study. Front. Endocrinol. https://doi.org/10.3389/fendo.2021.716681 (2021).
Google Scholar
-
Osei-Hyiaman, D., Ueji, M., Toyokawa, S., Takahashi, H. & Kano, K. Influence of grip strength on metacarpal bone mineral density in postmenopausal Japanese women: A cross-sectional study. Calcif. Tissue Int. 64(3), 263–266 (1999).
Google Scholar
-
Di Monaco, M., Di Monaco, R., Manca, M. & Cavanna, A. Handgrip strength is an independent predictor of distal radius bone mineral density in postmenopausal women. Clin. Rheumatol. 19(6), 473–476 (2000).
Google Scholar
-
Hasegawa, Y., Schneider, P. & Reiners, C. Age, sex, and grip strength determine architectural bone parameters assessed by peripheral quantitative computed tomography (pQCT) at the human radius. J. Biomech. 34(4), 497–503 (2001).
Google Scholar
-
Zimmermann, C. L., Smidt, G. L., Brooks, J. S., Kinsey, W. J. & Eekhoff, T. L. Relationship of extremity muscle torque and bone mineral density in postmenopausal women. Phys. Ther. 70(5), 302–309 (1990).
Google Scholar
-
Foley, K. T., Owings, T. M., Pavol, M. J. & Grabiner, M. D. Maximum grip strength is not related to bone mineral density of the proximal femur in older adults. Calcif. Tissue Int. 64(4), 291–294 (1999).
Google Scholar
-
Picca, A. et al. Bone-muscle crosstalk: Unraveling new therapeutic targets for osteoporosis. Curr. Pharm. Des. 23(41), 6256–6263 (2017).
Google Scholar
-
Sakuma, K. & Yamaguchi, A. Sarcopenia and cachexia: The adaptations of negative regulators of skeletal muscle mass. J. Cachexia Sarcopenia Muscle 3(2), 77–94 (2012).
Google Scholar
-
Wang, J., Leung, K. S., Chow, S. K. & Cheung, W. H. Inflammation and age-associated skeletal muscle deterioration (sarcopaenia). J. Orthop. Transl. 10, 94–101 (2017).
Acknowledgements
The authors acknowledged the GEnetic Factors for OSteoporosis Consortium, the UK Biobank and Breast Cancer Association Consortium for contributing the data used in this work.
Funding
This study was funded by Medical Research Project of Luzhou-Southwest Medical University (2019LZXNYDJ37), Scientific Research Cultivation Project of The Affiliated Traditional Chinese Medicine Hospital of Southwest Medical University (2022-CXTD-08), China Postdoctoral Science Foundation (2022M720602), Chongqing Special Postdoctoral Science Foundation (XmT2022072), Natural Science Foundation of Chongqing (cstc2019jcyj-msxmX0836).
Author information
Authors and Affiliations
Contributions
X.Q.M., B.H., M.Z.Z., Y.Z. and X.J.C. conducted literature search, data collection and statistical analysis. X.Q.M., B.H., Y.S.O. and X.J.C. conducted study design, data interpretation and manuscript preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Tables.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Reprints and Permissions
About this article
Cite this article
Mou, X., He, B., Zhang, M. et al. Causal influence of muscle weakness on cardiometabolic diseases and osteoporosis.
Sci Rep 13, 19974 (2023). https://doi.org/10.1038/s41598-023-46837-y
-
Received: 10 February 2023
-
Accepted: 06 November 2023
-
Published: 15 November 2023
-
DOI: https://doi.org/10.1038/s41598-023-46837-y
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.